|
Between-group differences in mean RMV were statistically significant at week 6 in both the primary ITT (P = 0.005) and OT (P = 0.006) populations. Sixty patients completed all 8 weeks of the study (on-treatment population): 91% (30) of the 33 patients in the test group, and 88% (30) of the 34 patients in the control group. The resulting primary intent-to-treat (ITT) population included 33 patients in the test group and 34 patients in the control group. Adverse events (AEs), including erythema, scaling, and edema, were also assessed by a dermatologist using the visual grading scale of Frosch and Kligman and COLIPA. Other skin properties, including moisture content, pH, and redness (erythema value), were measured. 
The assessments of melasma intensity and other skin properties were performed before administration (week 0) and every 2 weeks thereafter for up to 8 weeks. Additionally, the severity of melasma was determined visually using the Melasma Area and Severity Index (MASI) scored independently by 3 investigators. This parameter was used as a primary outcome of this study. Subjects were assessed for the intensity of their hyperpigmented skin area by measuring the difference in the absolute melanin value between hyperpigmented skin and normal skin (RMV). The supplemental sunscreen product with sun protection factor 30 was distributed to all patients. In this single-center, randomized, double-blind, controlled study, Thai patients with mild to moderate facial melasma (relative melanin value in range of 20-120) were randomized for the application of either the test or the emulsion-based (control) product in the morning and before bedtime for 8 weeks. The purpose of this study was to assess the efficacy of a formulation containing a combination of trans-4-(aminomethyl) cyclohexanecarboxylic acid/potassium azeloyl diglycinate/niacinamide compared with an emulsion-based control in the treatment of melasma in Thai adults. Treatment of melasma involves the use of topical hypopigmenting agents such as hydroquinone, tretinoin, and azelaic acid and its derivatives.  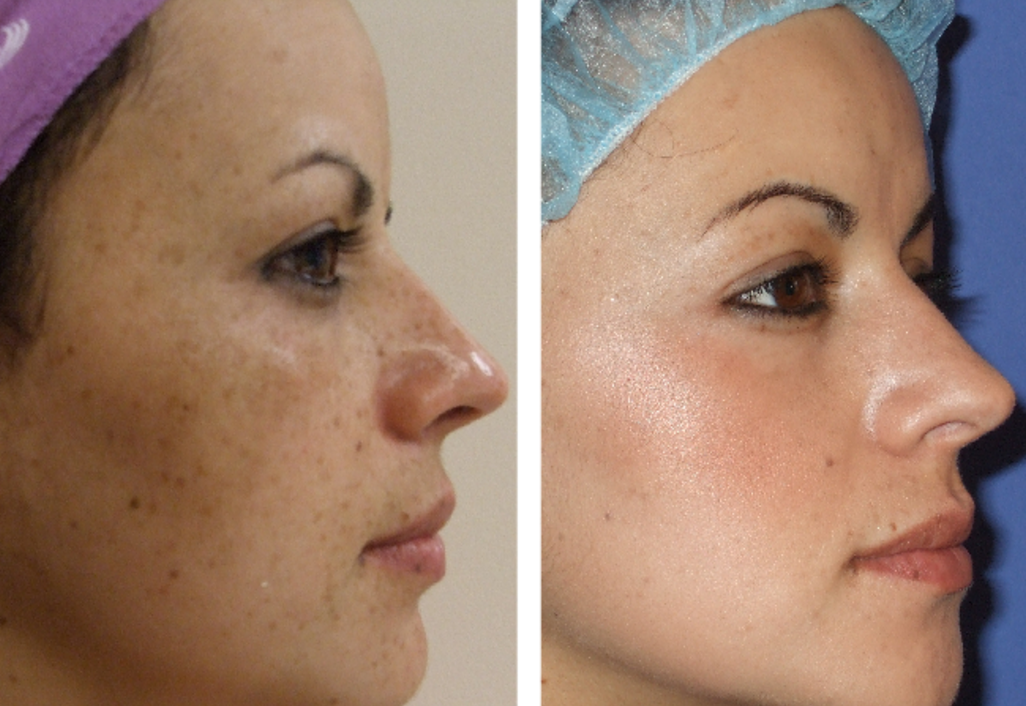
Melasma is an acquired hyperpigmentary disorder characterized by dark patches or macules located on the cheeks, forehead, upper lip, chin, and neck. 5 Cosmetics and Natural Products Research Center, Faculty of Pharmaceutical Sciences and Center of Excellence for Innovation in Chemistry, Naresuan University, Phitsanulok, Thailand Cosmetics and Natural Products Research Center, Faculty of Pharmaceutical Sciences and Center of Excellence for Innovation in Chemistry, Naresuan University, Phitsanulok, Thailand.4 Cosmetics and Natural Products Research Center, Faculty of Pharmaceutical Sciences and Center of Excellence for Innovation in Chemistry, Naresuan University, Phitsanulok, Thailand.3 Department of Pharmaceutical Technology, Faculty of Pharmaceutical Sciences and Center of Excellence for Innovation in Chemistry, Naresuan University, Phitsanulok, Thailand.2 Pazana Laboratory Asia Co., Ltd, Bangyikhan, Bangplad, Bangkok, Thailand.1 Department of Pharmaceutical Technology, Faculty of Pharmaceutical Sciences and Center of Excellence for Innovation in Chemistry, Naresuan University, Phitsanulok, Thailand Cosmetics and Natural Products Research Center, Faculty of Pharmaceutical Sciences and Center of Excellence for Innovation in Chemistry, Naresuan University, Phitsanulok, Thailand.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
